Updated: July 5, 2018
Labdoor contracted xxxxxxx*, an FDA-registered laboratory owned by xxxxxxx, to perform purity and label accuracy testing on 36 magnesium supplements. The results for the Natural Vitality Calm product are as follows:

Upon publishing our reports, we received an email from a Natural Vitality representative who wanted to see the Certificate of Analysis (CoA) for their product. We sent them the xxxxxxx CoA for their product (included at the end of this statement).
Unbeknownst to us, Natural Vitality has been a customer of xxxxxxx for nearly a decade. When they found out that these results were generated by xxxxxxx, they used their influence with xxxxxxx to breach our laboratory records and convince a xxxxxxx scientist to reinterpret their results, double the serving size, and recalculate mineral and heavy metal values. xxxxxxx recalculated results are as follows:
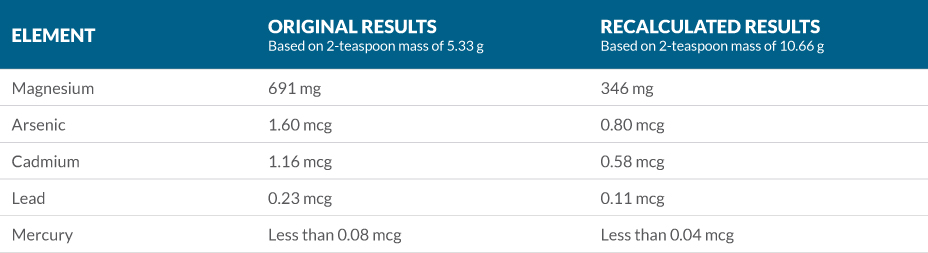
As you can see, this cuts all values in half since mineral and heavy metal values are generated on a per serving size basis. We see that magnesium content is much closer to its label claim of 350 mg per serving, with a slight underage. All heavy metal concentrations are also cut in half.
Even if we assume the higher sample mass of 10.66 g, this product would still receive a D+ grade on Labdoor due to high heavy metal content. For example, xxxxxxx recorded the arsenic content of the #1-rated LifeExtension Magnesium Caps at less than 0.03 mcg per serving vs. Natural Vitality Calm at 1.60 mcg per serving. Even using the most favorable serving size calculations, which would scale the arsenic content down to 0.80 mcg per serving, we’re talking about an over 25X difference in arsenic content. Consumers deserve to see this information, and that’s why we publish it.

It’s important to note that Labdoor is not claiming that this product fails California Proposition 65 limits. There is no official Maximum Allowable Dose Level (MADL) set by the Office of Environmental Health Hazards Assessment (OEHHA) for arsenic. Thus, we cited the OEHHA’s Proposition 65 draft MADL. That proposal recommends a 0.1 mcg per day limit for inorganic arsenic. This MADL recommendation has been stuck in draft format since 2003, but we wanted consumers to have all of the available information.
This isn’t a Proposition 65 issue (yet). This is a Labdoor issue. If a supplement is projected to exceed at least one of our four heavy metals limits, it will receive less than 5 points out of 10 in Labdoor’s Purity score. That’s our choice, and we believe it serves customers well to be able to easily gauge the relative purity of these health products.
Labdoor’s official reports and rankings will continue to be based upon xxxxxxx original Certificate of Analysis. We will not be using the revised results since they were generated due to laboratory interference by the company being reviewed. If you would like to use xxxxxxx revised results for the purposes of your own research, then that data is freely available in this statement. We also intend to retest a new sample of this product at a new independent laboratory. This retest will be based on a new lot of this product, so if the heavy metals concentrations in this product have improved, this will be reflected in our new rankings. The results of this third-party analysis will be final, and this data will be final for the purposes of all Labdoor reports.
Additionally, we have received a few messages from companies whose magnesium supplements were tested as part of this analysis who argued that arsenic contamination exists in many common food products, including rice, and thus is normal and should not be penalized.
Here are the facts: Different magnesium supplements tested at vastly different arsenic levels, ranging from less than 0.01 mcg per serving to more than 1.00 mcg per serving. We think this is important information for consumers to know when researching a supplement, and it is why we included it in our grading system.
If you have any questions about this analysis or any other research performed by Labdoor, we’re happy to answer your questions via email, Facebook, Twitter, or Reddit.
*Note: Labdoor has edited this article to anonymize the laboratory who performed this analysis at their request and in compliance with their Terms and Conditions.
